A £2 million hyperpolariser facility at Addenbrooke’s Radiology Department was officially opened today by John Dineen, president and CEO of GE Healthcare, who was accompanied by Dr Keith McNeil, CUHFT Chief Executive Officer and Professor Patrick Maxwell, the Regius Professor of Physic.
The SPINlab machine will allow clinicians to see molecular changes in cells as they happen in patients, opening up a vast range of potential applications of the technology for detecting cancerous cells and monitoring the immediate effects of treatment on tumours.
The new method of imaging could in future enable clinicians to see whether or not a drug is working on a tumour within a day or two of the patient starting treatment, instead of the current wait of several weeks for evidence of tumour shrinkage. This means that patients could stop taking ineffective drugs, try different ones and receive the best treatment for their cancer as quickly as possible. The other key benefit for patients is that the scanning process is relatively quick.
Scientists at the Cancer Research UK Cambridge Institute, led by Professor Kevin Brindle have, with colleagues from GE Healthcare, developed the revolutionary imaging technique known as nuclear spin hyperpolarisation. Pyruvate, a common compound found in all cells, is labelled with a special type of carbon, called carbon 13 (C-13). This is then hyperpolarised, which causes the carbon molecules to become at least 10,000 times more visible within a Magnetic Resonance (MR) scanner. This means that the C-13 pyruvate signals can be tracked as they move around the body and enter different cells, and clinicians can plot how quickly the compound is broken down and converted into new substances within the cell. This information is critical in assessing whether or not a drug is effective in killing cancer cells.
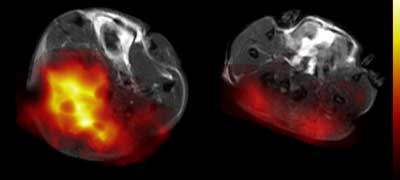
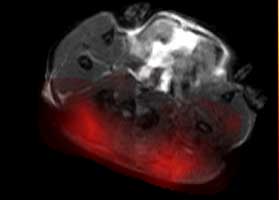
The false-coloured MRI image, below, shows a tumour on the left prior to drug treatment and, on the right, the same tumour 24 hours after treatment. The reduced coloured area reveals a rapid decrease in pyruvate metabolism following treatment, indicating that the drug is already starting to kill tumour cells.
After many years of research, in collaboration with a GE spin out company, Research Circle Technology, this technique is now being translated into the clinic with the production of the SPINlab equipment. There are currently just 5 SPINlab machines installed worldwide and Cambridge expects to be the second site, after San Francisco, to conduct studies in patients.
The research and development team has had to overcome many technical problems to reach this point. The C-13 pyruvate needs to be super-cooled and surrounded by an intense magnetic field to trigger the hyperpolarisation process. Once the substance is hyperpolarised, time is critical as the labelled carbon molecules lose their unique signal within a few minutes. The substance needs to be rapidly warmed up so that it can be injected into a patient already waiting in the MR scanner, so the scanning process can start without wasting valuable monitoring time.
The technology has already shown promising results in non-clinical trials and it has been tested on prostate cancer patients in the US. Clinical trials on cancer patients at Addenbrooke’s are due to start later this year.
Professor Brindle said, ‘We are hoping that it will lead to patients not having to take drugs that will not work for them. There are treatment choices – some people respond well to one treatment, and others don’t. The standard method for assessing whether a treatment has worked is for a patient to take a course of treatment and then determine whether the tumour has shrunk or not. This new technique could allow doctors to tell much more quickly whether a treatment is working.’
In recognition of the collective achievement of Professor Brindle’s Group, the team at the University of California in San Francisco and GE Healthcare in developing and producing the new technology, the World Molecular Imaging Society has awarded a prestigious Gold Medal for the teams involved in the project.
The facility and early phase clinical trials are funded and supported by Cancer Research UK, Wellcome Trust, Addenbrooke’s Charitable Trust, Cambridge Cancer Centre, GE Healthcare and the University of Cambridge.