Urological Malignancies Virtual Institute

Prostate and renal cancers are key contributors of cancer related death and morbidity. Early detection and optimal primary treatment improves cure rates and reduces progression to metastasis. Our clinicians and scientists have developed closely integrated multi-disciplinary research themes including; exploring new markers and imaging for early detection, better modelling of early disease biology, improved methods of risk stratification and novel therapeutic window studies alongside standard clinical treatments. This work is significantly enhanced by our large bio-repository and high-volume urological cancer practice in Addenbrooke's.
The Urological Malignancies Virtual Institute has a new online home - cambridge-urologicalmalignancies.org.uk. The new website includes a blog and trials information for fellow clinicians, researchers and the general public, as well as resources for Virtual Institute members.
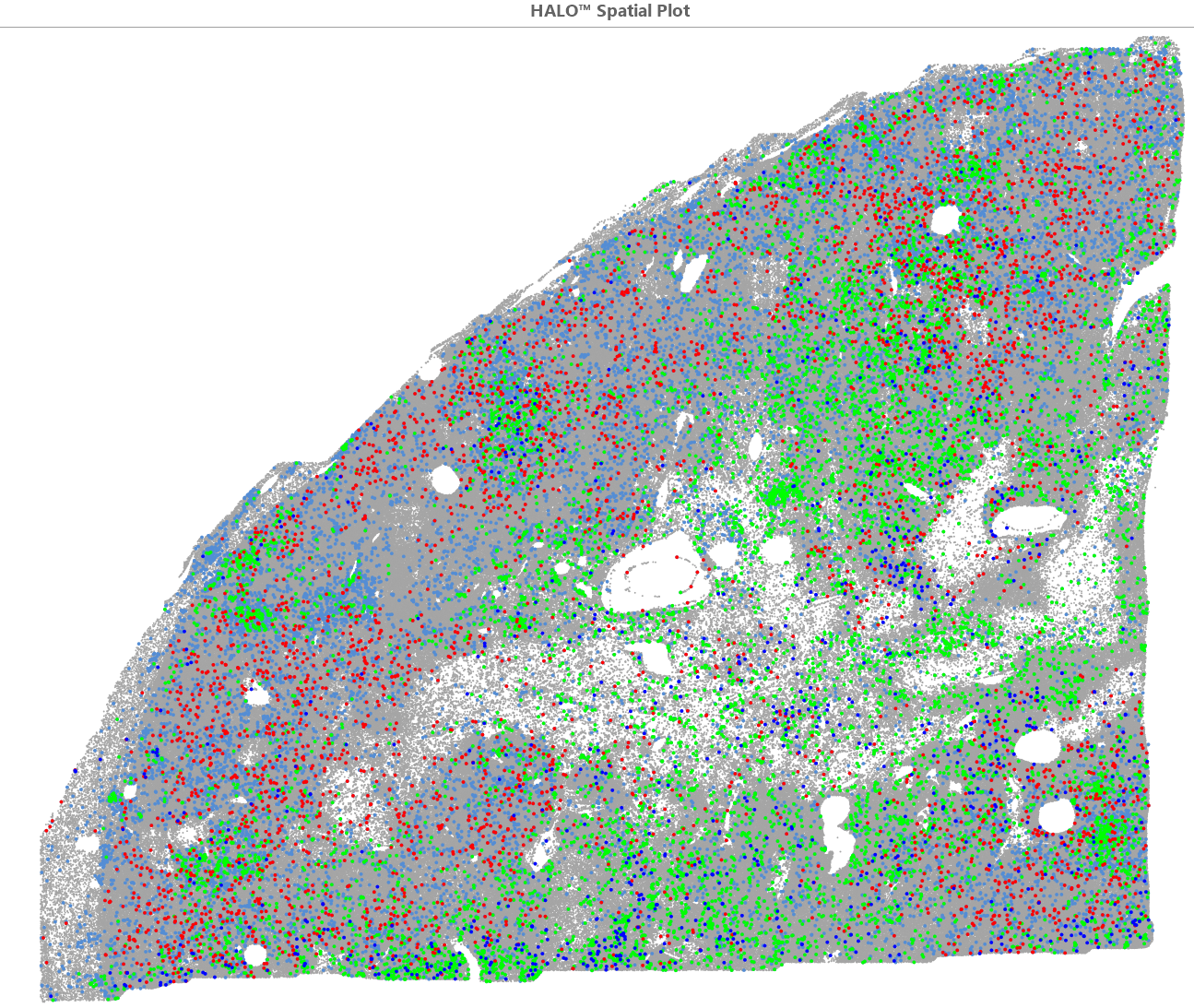
The Cambridge Renal Cancer Group (CamRenCan) is a collaboration of basic and translational scientists together with clinicians working synergistically towards the common goal of improving outcomes of patients with renal cancer via multimodal research strategies.
The Centre runs internationally recognised programmes in the genetics of the disease and the metabolic consequences of those genetic abnormalities, in understanding tumour biology and in the treatment of resulting malignancies.
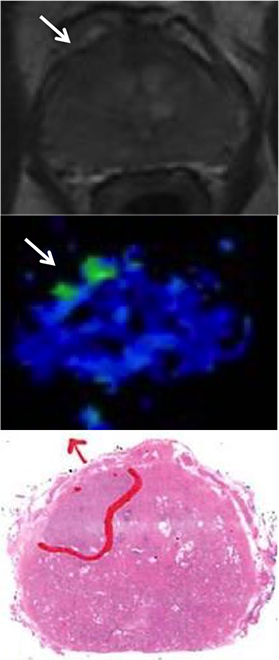
We are a UK centre for major national and international studies including ProtecT, CANCAP (lead) VoxTox (lead), ICGC (major contributor), PRACTICALS, MOVEMBER, Genomic England, STAMPEDE, RADICALS, PACE, and NIHR IHC. In the last 5 years the prostate research group has together published over 200 papers and initiated 8 translational investigator led studies.
The current research focus in Cambridge is on improving the management and outcomes of men presenting with non-metastatic prostate cancer (over 80% of all diagnosis).



















