Clinical research on prostate cancer
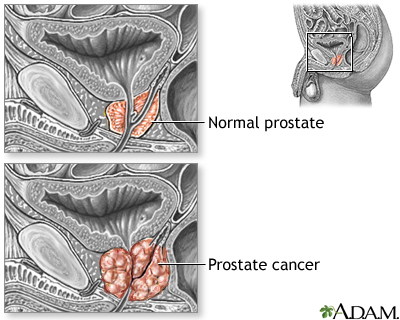
Prostate cancer is the most common cancer in men with 40,000 new cases each year and 10,000 deaths in the UK. In adults, prostate cancer is the second most common cancer. The type of prostate cancer depends on the type of cell that the cancer started in. The most common type are adenocarcinomas. Other types include transitional cell (or urothelial) cancer and this starts in the cells that line the urethra, which carries urine to the outside of the body but this is very rare. Other rarer cancers include squamous cell cancer, small cell prostate cancer, carcinoid and sarcomas.
Treatment for prostate cancer may not happen straight away, instead Doctors will monitor the cancer closely. Depending on the situation, they call this active surveillance or watchful waiting. If the prostate cancer is to be treated then the main treatments include surgery, external radiotherapy, brachytherapy (internal radiotherapy), hormone therapy, high frequency ultrasound therapy (HIFU), cryotherapy, chemotherapy and symptom control treatment. Patients may have one or more of these treatments depending on the stage of the cancer and whether the cancer is low risk, medium risk or high risk.
Survival depends on many different factors including the individual's condition, type of cancer, treatment and fitness level. For men with prostate cancer in England and Wales, around 95% will survive their cancer for 1 year or more, almost 90% will survive their cancer for 5 years or more and more than 80% will survive their cancer for 10 years or more.
Prostate cancer is often slow-growing, but more aggressive forms of the disease are hard to treat. Scientists and clinicians in Cambridge are working together to understand the molecular changes that occur before and during the early stages of prostate cancer.
As part of a comprehensive high quality surgical service, Addenbrooke’s Hospital runs the largest prostate cancer surgery service in the UK. Robotic surgery has reduced average in-patient stays from six days to one and results in much improved quality of life following surgery. Almost all prostate operations are carried out using this technique at Cambridge, compared to a national average of 20%.
Click the 'Find out more' button to view a short film presenting a profile of the urology team.
Cambridge has the only Biopsy MRI-Transrectal fusion (Heidelberg) machine in the UK which overlays MRI images with ultrasound guided prostate biopsies. We are currently working to incorporate PET imaging with testing of different tracers and MRI to non-invasively detect response to treatment in high grade tumours. We are using imagery to monitor the response to new drugs in trials on men about to have radical surgery.
The largest randomised control trial of surgery, radiotherapy and active monitoring of localised prostate cancer, which will produce a risk-based gold standard of management.
ProtecT is the largest ever randomised clinical trial of over 100,000 participants funded by the National Institute for Health Research (NIHR). It is being run by the Cambridge Cancer Centre, Oxford University and Bristol University. The trial is assessing the effectiveness of surgery, monitoring and radiotherapy in screen-detected prostate cancer. The associated collection of tissue samples is being amassed in the Cambridge Cancer Centre.
We are carrying out studies of the protein receptor for the male sex hormone, the androgen receptor (AR), which is critical to prostate cancer development and growth. We have identified AR regulated genomic signatures that predict outcomes, which is exciting because it may be possible to identify which men are likely to progress after radical treatment and how to target aberrant molecular pathways to improve treatment. We have developed the concept of ‘window trials’, which are studies done using imaging and biological end-points in men treated with short courses of novel drugs prior to surgery or radiotherapy. These studies are important for patients, science and for pharmaceutical companies.
The table below lists the current open trials for prostate cancer coordinated by the Cambridge Cancer Trials Centre. If you would like further information about how to take part in any of the clinical trials listed here, please talk to your cancer specialist as patients usually need to be referred by their doctor




















