Scientific Research Groups
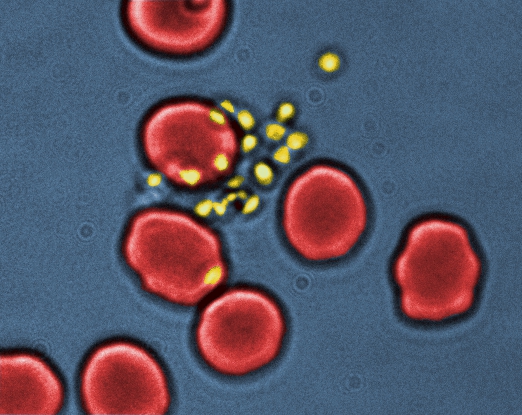
Pietro Cicuta is a Professor of Biological Physics in the Physics Department, Cambridge University. His team combines: (a) experience in single cell measuring on micro-organisms, including studies of cell size, adhesion, gene regulation; (b) microfluidic fabrication and automation of imaging and optical measurements; (c) quantitative mechanistic/physical modelling. Developing new experimental techniques to study the mechanics and dynamics of soft and biological systems is the heart of his research. Recent work includes studies of model bacteria and pathogens in host/pathogen interactions, physiology of ciliated epithelia and mechanics/lipidomics of cell membranes.
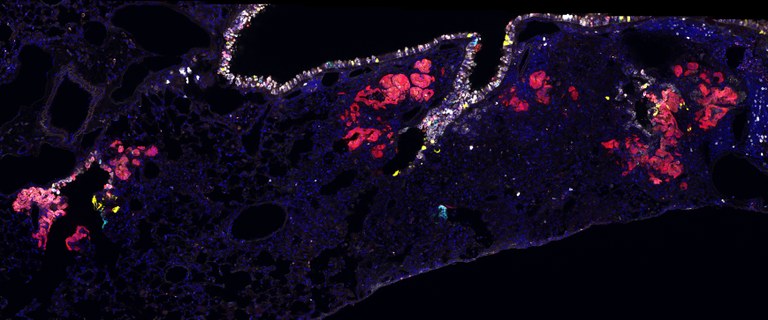
What are the regulatory mechanisms that control homeostatic turnover, and how do their perturbation contribute to disease progression? The lung is a very slow cycling organ that is composed of diverse epithelial and stromal cell types, but has capacity to rapidly regenerate new cells after injury. Lee group is trying to understand how stem cells respond to different signals from their local environment and orchestrate the changes in chromatin, transcription, translation, and cellular dynamics in homeostasis and injury repair. We investigate the regulatory networks that need to be turned on and off at the right time and place for stem cells to become activated and generate specialised cell types during regeneration. We are also interested in defining cellular heterogeneity and plasticity during this process. Elucidating the normal process of lung dynamics will provide us a foundation to understand lung diseases and cancer. We couple ex vivo 3D organoid cultures of human and mouse lungs with genetic tools, in vivo transgenic mouse models with lineage tracing techniques, quantitative mathematic modelling of clonal dynamics, and bioinformatics at the single cell level.
Ultan McDermott's group is based at the CRUK Cambridge Institute and is focused on understanding drug resistance in cancer, using CRISPR technologies and single cell sequencing.
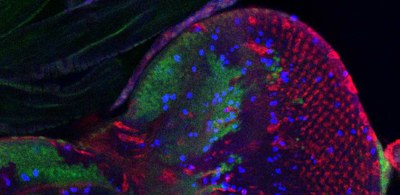
Professor Stefan Marciniak's lab wish to understand the cellular consequences of endoplasmic reticulum stress; in particular its effects on tissue growth and cell survival. In doing so, they hope to identify targets for the development of novel therapies.
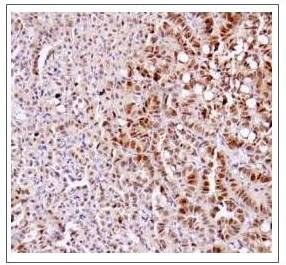
The main aim of Carla Martin's programme is to define the molecular and functional alterations that enable lung tumour progression from benign stages to high-grade adenocarcinoma. While their immediate goal is to improve the targeting of tumours harbouring particular sets of mutations, their ultimate goal is to identify broader and targetable signatures associated with benign and advanced disease. They are currently focused on the evolution of lung tumours expressing mutant versions of Kras and p53.
Dr Daniel Muñoz-Espín is based at the Department of Oncology within the University of Cambridge School of Clinical Medicine. His group focuses on the fundamental processes and mechanisms related to lung cancer initiation and progression. These processes and cellular estates include, among others, senescence, cellular plasticity, cancer stemness and their contribution to the tumour microenvironment. By using 2D/3D cultures, mouse models of lung carcinogenesis and human tissue samples Muñoz-Espín´s team investigates what are the key factors, signalling pathways, gene expression profiles, and genetic and epigenetic landscapes promoting cancer. Elucidating how cellular damage and oncogene activation insults can result in the malignant transformation of differentiated cells, and how cancer cells relate to the tumour microenvironment is crucial to develop more efficient strategies and sophisticated translational tools for precision medicine. Therefore, Daniel and his team use this knowledge for the preclinical design, development and validation of novel tools for cancer early detection and therapeutic interventions. Their ultimate goal is to translate these innovative strategies into early phase clinical trials.

Emma Rawlins' lab - based at the Gurdon Institute, works on the roles of stem cells in lung development and homeostasis with the dual aims of understanding how the normal homeostatic control mechanisms are subverted in cancer and whether mechanisms of differentiation can be exploited as possible therapies. They investigate these questions using both in vivo mouse models and human organoids.



















