Dr Madeline Lancaster
Position: Group Leader
Personal home page:
https://www2.mrc-lmb.cam.ac.uk/groups/lancaster/
PubMed journal articles - click here
Dr Madeline Lancaster is pleased to consider applications from prospective PhD students.
The human brain is highly unique in both its size and complexity. Human brain size is greatly expanded compared with even our closest living relatives, chimpanzees and gorillas. On top of this, the greatly expanded number of neurons in the human brain must properly wire up to make the functional brain connectome that is at the core of our unique cognitive abilities. Despite these crucial differences in humans compared with other animals, we know very little about the mechanisms that lead to the highly enlarged and complex connectivity of the human brain.
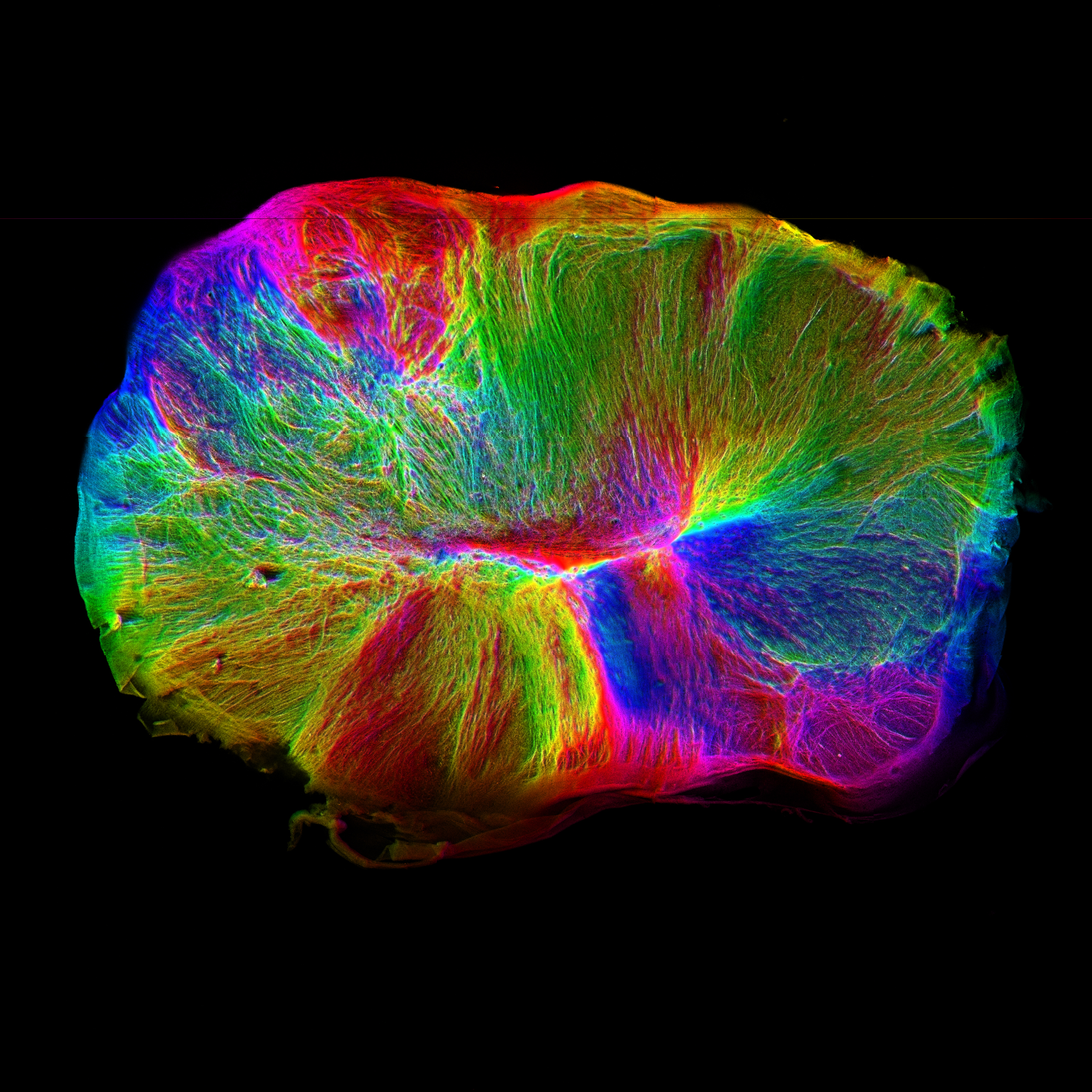
In order to explore these questions, we use in vitro models called cerebral organoids, which are self-organizing stem cell-derived tissues that model many aspects of the developing brain. We perform a combination of evolutionary and developmental studies to determine the molecular mechanisms that control brain size and neuronal connectivity. Our findings are revealing a number of key cell and molecular biological principles that set the stage for development of the complex human brain. In particular, cell shape, physical forces, and key developmental signaling pathways can promote or delay fate transitions that guide the production of neurons. We are also exploring the roles of supportive cell types such as the cerebrospinal fluid-producing choroid plexus. Finally, we are examining the development of neuronal connectivity in these systems, and looking at how network topology is established and remodeled in the developing human brain.
Through this combination of approaches, we hope to contribute to an important area of human health, namely understanding human brain development. Because neurological conditions affect features that are quintessentially human, a better understanding of the development of our brain will be a vital part of understanding what aspects go awry in diseases, particularly neurodevelopmental disorders. We also hope to understand how the mechanisms that control brain size may similarly be involved in neural overgrowth as seen in brain cancer.
Symplectic Elements feed provided by Research Information, University of Cambridge
Pellegrini L, Bonfio C, Chadwick J, Begum F, Skehel M, Lancaster MA. Human CNS barrier-forming organoids with cerebrospinal fluid production. Science. 2020 Jul 10;369(6500):eaaz5626.
Giandomenico SL, Mierau SB, Gibbons GM, Wenger LMD, Masullo L, Sit T, Sutcliffe M, Boulanger J, Tripodi M, Derivery E, Paulsen O, Lakatos A, Lancaster MA. Cerebral organoids at the air-liquid interface generate diverse nerve tracts with functional output. Nat Neurosci. 2019. Apr;22(4):669-679.
Kelava I, Lancaster MA. Stem cell models of human brain development. Cell Stem Cell. 2016 Jun 2;18(6):736-48.
Lancaster MA^, Corsini NS, Wolfinger S, Gustafson EH, Phillips AW, Burkard TR, Otani T, Livesey FJ, Knoblich JA^. Guided self-organization and cortical plate formation in human brain organoids. Nat Biotechnol. 2017 May 31.
Renner M*, Lancaster MA*, Bian S, Choi H, Ku T, Peer A, Chung K, Knoblich JA. Self-organized developmental patterning and differentiation in cerebral organoids. EMBO J. 2017 May 15;36(10):1316-1329.
Luo C*, Lancaster MA*, Castanon R, Nery JR, Knoblich JA, Ecker JR. Cerebral Organoids Recapitulate Epigenomic Signatures of the Human Fetal Brain. Cell Rep. 2016 Dec 20;17(12):3369-3384.
Lancaster MA^,Knoblich JA^. Generation of cerebral organoids from human pluripotent stem cells. Nat. Protoc. 2014, 9(10): 2329-40.
Lancaster MA,Knoblich JA. Organogenesis in a dish: Modeling development and disease using organoid technologies. Science. 2014, 345(6194):1247125.
Lancaster MA, Renner M, Martin C-A, Wenzel D, Bicknell LS, Hurles ME, Homfray T, Penninger JM, Jackson AP, Knoblich JA. Cerebral organoids model human brain development and microcephaly. Nature. 2013, 501(7467):373-9.